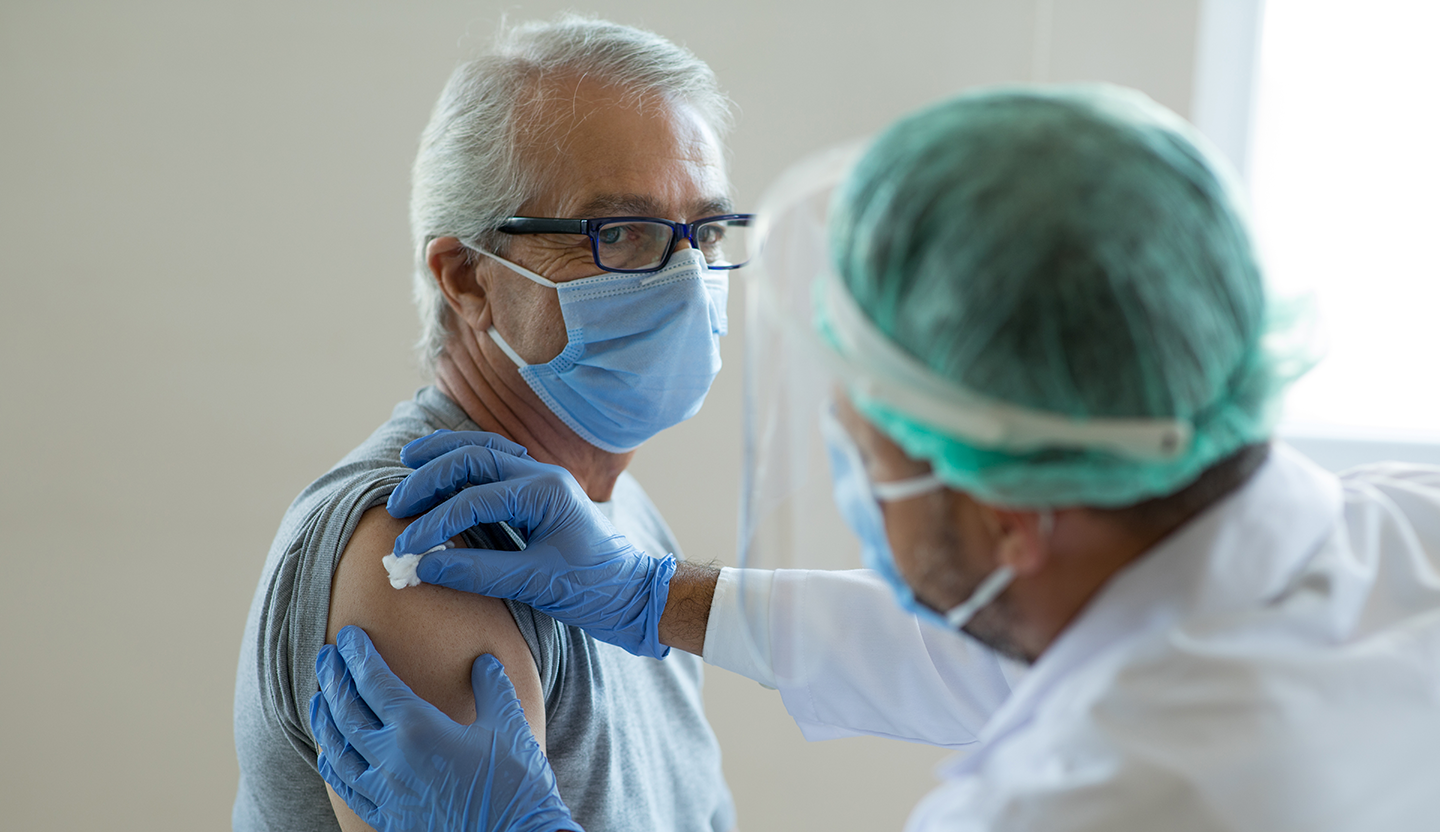
Explore the latest evidence-based information about vaccines approved for use to prevent COVID-19 infections. View vaccine drug monograph information as well as patient education resources.
ClinicalKey
Note:
User acknowledges that these COVID-19-related materials are provided for use at no charge as part of Elsevier’s efforts to support healthcare providers and patients. The information contained within the website is intended as a supplement to, and not a substitute for, the knowledge, expertise, skill and judgment of healthcare professionals involved in patient care. The content within this page may not be re-used, modified or re-distributed without prior written permission.
Cookies are used by this site. To decline or learn more, visit our cookie notice.
Copyright © 2025 Elsevier, its licensors, and contributors. All rights are reserved, including those for text and data mining, AI training, and similar technologies.